
Cal's Angels Advanced Leukemia Clinical and Research Program
Impact Update — October 2025
With the support of committed donors like you, Cal’s Angels Advanced Leukemia Clinical and Research Program is transforming the outlook for young people diagnosed with aggressive leukemias. Your generosity has enabled our multidisciplinary team of specialists to not only care for the most complex cases we see today, but also to pioneer the treatments of tomorrow. Because of your generosity, we are giving families hope, advancing science and rewriting what is possible.
With gratitude, we are pleased to share how your philanthropy has elevated our ability to serve every child and family in need of expert care.
Delivering Cutting-Edge Clinical Care

Developing more effective treatments for aggressive cancers requires relentless dedication to understanding their complexities and exploring new approaches. Our team, led by Drs. Jenna Rossoff and Sara Zarnegar Lumley, is committed to ensuring our young patients have access to the latest research-informed therapies that give them the best chance to overcome their disease. We participate in all major early-phase pediatric clinical trial networks and collaborate with pharmaceutical industry partners to offer cutting-edge treatments unavailable elsewhere in Illinois or the surrounding area.
Since July, we have opened two Phase 1/2 trials, and four patients were screened for or enrolled on these early phase trials. We saw eight patients with leukemia who were referred to our program for a second opinion. Of these, one patient was referred for CAR T-cell immunotherapy at Lurie Children’s, another was referred for CAR T-cell immunotherapy elsewhere and three patients were enrolled on early phase clinical trials at Lurie Children’s. The three remaining patients had complex relapse and received patient-tailored regimens at Lurie Children’s.
Key Program Measures
Q3 2025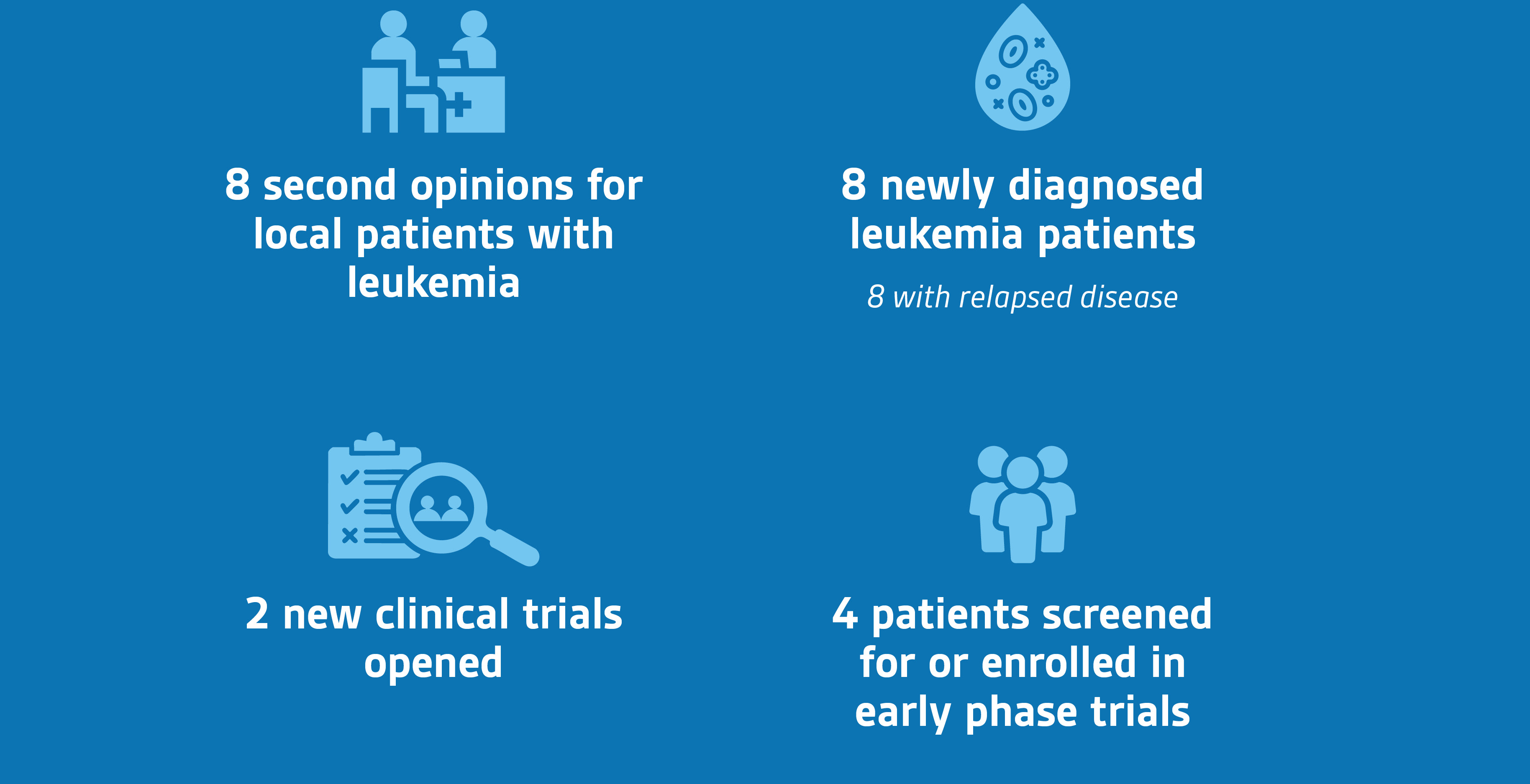

Growing our Team
We are excited to share that Lindsay Schwartz, MD, has joined the hematological malignancy team. Dr. Schwartz is a pediatric oncologist and physician-scientist whose research focuses on survivorship, fertility and rare non-Hodgkin lymphoma in young cancer patients. She trained at UCLA and the University of Chicago. Her work has earned national recognition, including awards from The Society for Industrial and Organizational Psychology and The American Society for Pediatric Hematology/Oncology. Although funding from Cal's Angels did not support Dr. Schwartz's recruitment, our patients will benefit from the expertise she brings to our team.
Accelerating Novel Research
With support from Cal's Angels, Xiaopei “Lily” Zeng, MD, continues to study ways to improve the efficacy of targeted cellular and immunotherapies in the treatment of high-risk leukemias. Specifically, she aims to understand how B-acute lymphoblastic leukemia (B-ALL) evades targeted immunotherapy, including CD19 CAR-T cell therapy, to overcome this resistance.

Dr. Zeng’s earlier research revealed pathways related to cellular metabolism to be significantly different between cells that have downregulated CD19 expression after CD19 CAR T exposure compared to those with retained CD19 expression. To explore this further, she compared the metabolic profiles of cells that lost CD19 expression to those that retained it, identifying key differences in their chemical makeup. These findings are guiding her next steps, which include testing how specific metabolic pathways influence treatment resistance. Furthermore, she plans to use publicly available datasets of patient B-ALL sequencing data to strengthen the connection between lab findings and real-world outcomes.
We also have opened two new clinical trials:
- A Phase 1/2 trial of trametinib with risk-stratified chemotherapy for patients with newly diagnosed juvenile myelomonocytic leukemia
- A Phase 1/2 study of a novel prodrug OBI-3424 (OBI-3424) that is activated by a protein that is highly expressed in patients with relapsed/refractory T-cell acute lymphoblastic leukemia (T-ALL) and T-cell lymphoblastic lymphoma (T-LLy)

With Gratitude
We are very grateful for your longstanding commitment to our mission. Until the day childhood cancer is eradicated, our team will continue to work tirelessly to ensure our patients and families receive the highest level of comprehensive care. Thank you for all your support has made possible.