

Cal's Angels Advanced Leukemia Clinical and Research Program
Impact Update — July 2025
Thanks to your generous investment, Cal’s Angels Advanced Leukemia Clinical and Research Program provides cutting edge treatments and expert resources for pediatric, adolescent and young adult patients diagnosed with the most complex and challenging leukemias. We are excited to share that, in the past quarter, the program has made significant strides. We have expanded access to targeted therapies, reached more patients and enhanced patient-family support services.
With your partnership, we look forward to forging new paths in leukemia care and research for the children and families we serve.
Delivering Cutting-Edge Clinical Care
 From the initial diagnosis to follow-up, our experts work to provide seamless care and find the best possible treatment options specific to each child.
From the initial diagnosis to follow-up, our experts work to provide seamless care and find the best possible treatment options specific to each child.
Our team, led by Drs. Jenna Rossoff and Sara Zarnegar Lumley, is experienced in delivering therapies designed to improve survival and quality of life. Through collaborations with national cancer research consortiums and pharmaceutical industry partners, we can offer the very latest leukemia treatments being studied. These include phase 1 clinical trials, which many hospitals do not offer. These early studies are especially important for children with high-risk leukemia as they could offer more effective or less toxic alternatives to existing treatments.
Since April, we have initiated two new Phase 1 trials, and four patients were screened for or enrolled on early phase trials. We also launched our first inter-disciplinary tumor board where we reviewed referred patients to establish a treatment plan and next steps for care.
Key Program Measures
Q2 2025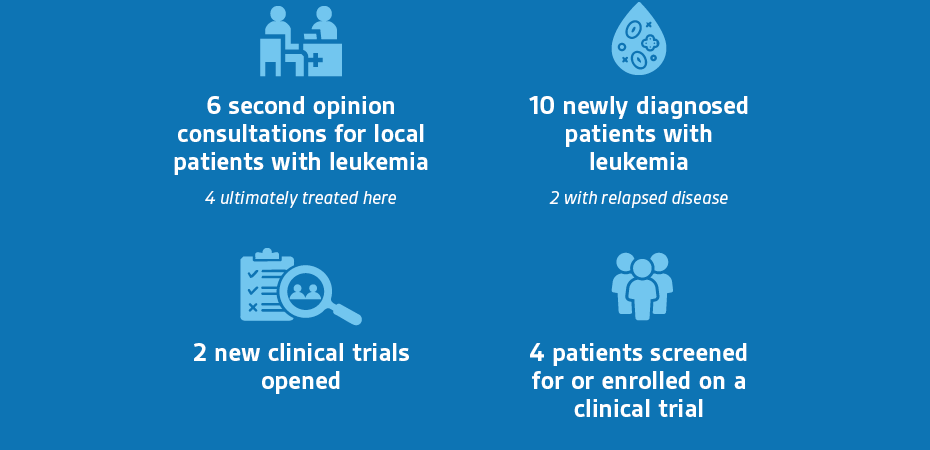
Building our Team
We recognize that a full spectrum of services—from leading edge medical treatment to compassionate, supportive services—are necessary to ensure our young patients can grow, develop and achieve their full potential. Your commitment will strengthen the level of comprehensive care we provide by allowing us to recruit a psychologist to address the psychosocial impact of cancer.
Mental health interventions delivered by a dedicated psychologist can give children the emotional tools they need to cope with uncertainty, stress and trauma. Whether helping a child process their feelings, guiding them through relaxation techniques or simply offering a safe space to express their fears, psychosocial support is essential to building resilience. Moreover, recognizing the first signs of emotional distress can lead to early intervention to minimize the negative impact on a child and ensure better long-term outcomes.
We are actively recruiting a pediatric psychologist to serve our patients with advanced leukemias and look forward to keeping you apprised of our progress.

Making a Difference with Your Support
Cal’s Angels Advanced Leukemia Clinical and Research Program is transforming care for young people diagnosed with aggressive leukemias. We are profoundly grateful for your vision and commitment, which have made this work possible. Thank you for your partnership.