
Cal's Angels Advanced Leukemia Clinical and Research Program
Impact Update — April 2025
Thanks to your generous investment, the Cal's Angels Advanced Leukemia Clinical and Research Program has launched under the leadership of oncologists Jenna Rossoff, MD, and Sara Zarnegar-Lumley, MD. The program offers specialized expertise and resources to care for pediatric, adolescent and young adult patients with high-risk subsets of leukemia, helping to fill a significant need for young people in Chicago and across the region. We are deeply grateful for your vision and commitment to bring this program to fruition.
We are pleased to share this report highlighting the significant progress made over the past three months. Thank you for partnering with us in this critical work.
Delivering Cutting-Edge Clinical Care
Our team is committed to ensuring young people who are diagnosed with leukemia today have access to the latest research-informed therapies that give them the best chance to overcome their disease and live to their fullest potential. Early phase pediatric clinical trials are crucial for advancing care and improving outcomes for children because they provide access to cutting-edge treatments not yet available to the broader public. Our institution has strong collaborations with major national research networks and pharmaceutical industry partners that will continue to grow under the infrastructure of the new Cal's Angels Advanced Leukemia Clinical and Research Program. Your support is enabling Dr. Zarnegar-Lumley to dedicate protected time as the operational lead for early phase trials. She is helping to optimize our workflow to support the expansion of Phase 1 and 2 clinical trials for leukemia.
Since January, we have initiated two new Phase 1 trials, and three patients were screened for or enrolled on early phase trials.
- A 12-year-old patient who recently completed treatment for B-ALL in May 2024 and relapsed in January 2025. The patient was enrolled on Phase 1 clinical trial of a novel cell death modulator with chemotherapy in partnership with the Dana Farber Cancer Institute. Since she is receiving treatment close to her home in northwest Indiana, the patient and her family (including two older brothers) can continue their daily work and school activities surrounded by their extended support network. She will continue to receive immunotherapy and a stem cell transplant at Lurie Children’s.
- A 2.5-year-old with refractory acute myeloid leukemia (AML) who was being treated at Comer Children’s Hospital. The patient was screened for a Phase 1 clinical trial through the Leukemia Lymphoma Society Pediatric Acute Leukemia Initiative. Our consultation found that the patient was in remission and ineligible for the clinical trial. He was able to return to his local hospital for stem cell transplant.
- A 4-year-old patient with refractory AML who was initially treated at Lurie Children’s but transferred to Comer in 2023 for a clinical trial. He is now returning for a Phase 1 clinical trial through the Leukemia Lymphoma Society Pediatric Acute Leukemia Initiative and will be the first pediatric patient enrolled on this international trial.
Many stringent requirements must be met for patients to participate in these demanding trials. Enrollment numbers can vary depending on patient eligibility and the evolving status of ongoing studies, as trials progress or meet their enrollment goals.
Key Program Measures
Q1 2025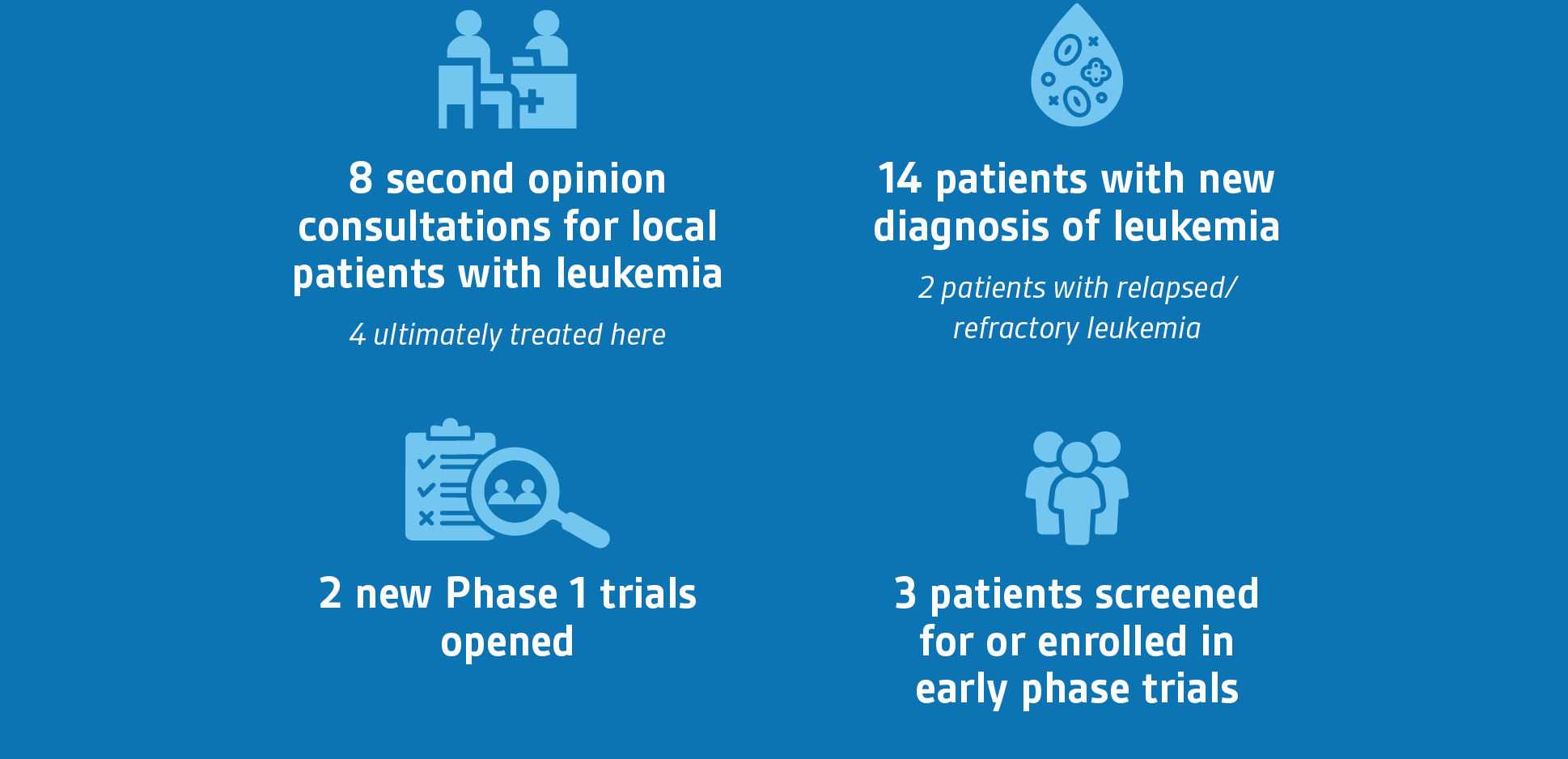
Building our Team
Thanks to your investment, we are becoming better equipped to accelerate groundbreaking clinical research and improve outcomes for young patients facing leukemia. Your support made it possible to add two new core team members:
Mikhail “Mik” Kuczmarski was hired as Clinical Trial Budget and Contract Manager, a role that is vital in advancing early-stage clinical trials for leukemia. By managing complex budget negotiations, ensuring contracts are efficiently executed, and maintaining compliance with regulatory processes, Mik helps move new therapies from concept to reality as quickly as possible. The manager’s expertise in navigating financial requirements, coordinating with pharmaceutical partners and supporting faculty ensures that promising clinical trials can launch without unnecessary delays—bringing hope to families seeking new options for their children. Mik previously served as Manager of Clinical Research Administration for WCG Clinical, a leader in the clinical trials industry that connects clinical research sponsors, CROs, sites and participants.
Jesenia “Jessie” Nordengreen was hired from a competitive pool of applicants as a Clinical Services Coordinator. In this patient facing role, Jessie will coordinate external patient referrals, obtain outside medical records and review insurance authorization, as well as schedule physician consultations, procedures and other appointments. Additionally, Jessie will schedule and facilitate monthly tumor board meetings and establish and maintain a database of patients seen by program physicians. She will also serve as the liaison to Lurie Children’s marketing department to ensure patients and providers in the community are aware of the program services and available trials. Jessie has seven years of experience in the Center for Cancer and Blood Disorders at Lurie Children’s. She is fluent in Spanish, which will allow for effective and welcoming interactions with patients/families with preferred language of English and Spanish as well as with other patients with the use of Interpreting Services.
Accelerating Novel Research
Breakthrough research leads to cutting-edge care. Your generosity will help to advance novel studies led by physician-scientists who are tirelessly seeking new and better ways to treat children with high-risk leukemia. We are pleased to share that Xiaopei “Lily” Zeng, MD, is the first researcher to benefit from Cal’s Angels funding.
Dr. Zeng is an Attending Physician in the Division of Hematology, Oncology, Neuro-Oncology & Stem Cell Transplantation and a research scientist in laboratory of Bin Zhang, MD, PhD, in the Robert H. Lurie Comprehensive Cancer Center of Northwestern University. Her overall research interest lies in understanding how B-acute lymphoblastic leukemia (B-ALL) evades targeted immunotherapy including CD19 CAR-T cell therapy. In the project supported by Cal’s Angels, Dr. Zeng will aim to better understand how changes in cellular metabolism pathways lead to resistance to CD19 targeted therapy. Her experiments include patient samples before and after receiving CAR-T cell therapy and metabolomic and genomic profiling to identify the factors that contribute to relapse after these therapies. We look forward to providing future updates as her work progresses.

With Gratitude
Your investment is shaping the future of pediatric leukemia care. By funding key initiatives in research and clinical care, you are not only improving the lives of the patients we serve today but also driving discoveries that will benefit children for generations to come. With your partnership, we remain committed to leading the field forward to bring hope to families who need it most.